Love Your Liver: Natural Ways to Keep It Strong for Life
Your liver is one of the hardest-working organs in your body, responsible for detoxifying harmful substances, aiding digestion, and regulating metabolism. Yet, it’s often overlooked—until problems arise. With rising cases of Fatty Liver Disease and Hepatitis, taking proactive steps to protect your liver is more important than ever. The good news? You can support your liver naturally with simple lifestyle changes.
Why Liver Health Matters
The liver performs over 500 vital functions, including filtering toxins from your blood, producing bile for digestion, and storing essential nutrients. When your liver is overburdened, it can lead to serious conditions like Cirrhosis or even liver failure.
Early damage often shows no symptoms, making prevention the most effective strategy for maintaining long-term health.
Top Natural Ways to Keep Your Liver Strong
1. Eat a Liver-Friendly Diet
A balanced diet is your first line of defense against liver disease. Focus on:
- Fresh fruits and vegetables (rich in antioxidants)
- Whole grains and fiber-rich foods
- Healthy fats like nuts and seeds
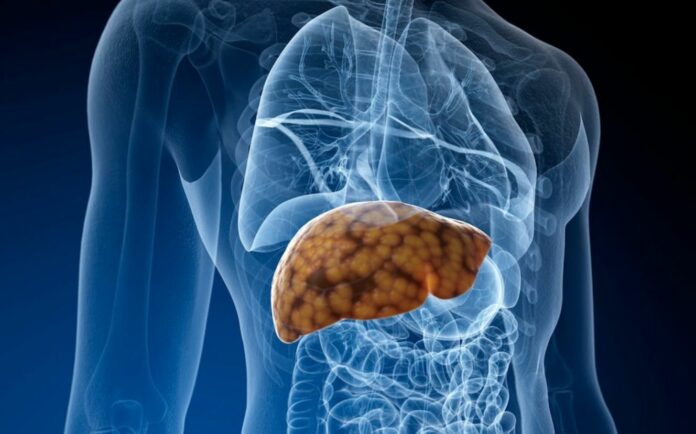
Avoid processed foods, excess sugar, and saturated fats, which can contribute to Nonalcoholic Fatty Liver Disease.
2. Stay Hydrated
Water helps flush toxins out of your body and supports liver function. Aim for at least 7–8 glasses daily to keep your system clean and efficient.
3. Limit Alcohol Consumption
Excess alcohol is one of the leading causes of liver damage. Over time, it can lead to conditions like Alcoholic Liver Disease. If you drink, do so in moderation—or avoid it altogether for optimal liver health.
4. Maintain a Healthy Weight
Being overweight increases your risk of developing fatty liver disease. Regular exercise, such as Brisk Walking or yoga, can help you maintain a healthy weight and improve liver function.
5. Avoid Toxins and Harmful Chemicals
Every day exposure to chemicals—from cleaning products to pesticides—can strain your liver. Try to:
- Use natural or eco-friendly products
- Wash fruits and vegetables thoroughly
- Avoid unnecessary medications
6. Use Medications Wisely
Overuse of certain medications, including Paracetamol, can harm your liver. Always follow dosage instructions and consult a doctor when in doubt.
7. Get Vaccinated
Vaccination is a powerful way to protect your liver from infections like Hepatitis A and Hepatitis B. Speak to your healthcare provider about recommended vaccines.
8. Include Liver-Supporting Foods
Certain foods are known to boost liver health naturally:
- Garlic (helps detoxification)
- Turmeric (anti-inflammatory properties)
- Green tea (rich in antioxidants)
- Leafy greens (support cleansing)
These foods can help reduce inflammation and improve liver function over time.
Signs Your Liver Needs Attention
Even though liver disease can be silent, watch for these warning signs:
- Fatigue and weakness
- Yellowing of the skin (jaundice)
- Abdominal pain or swelling
- Dark urine or pale stool
If you notice these symptoms, consult a healthcare professional immediately.
Simple Daily Habits for Long-Term Liver Health
Small lifestyle changes can make a big difference:
- Start your day with warm lemon water
- Stay physically active
- Get enough sleep (7–8 hours)
- Manage stress through meditation or yoga
Consistency is key when it comes to protecting your liver.
Conclusion
Your liver plays a crucial role in keeping your body healthy and functioning smoothly. By adopting natural, healthy habits, you can prevent liver disease and improve your overall well-being. From eating a balanced diet to staying active and avoiding toxins, every small step counts.
Love your liver today—because a healthy liver means a healthier, longer life.
References:
https://www.medicalnewstoday.com/articles/fatty-liver
https://www.healthline.com/nutrition/11-foods-for-your-liver
https://www.prevention.com/food-nutrition/a64577852/foods-good-for-liver/
https://www.hims.com/guides/how-to-improve-liver-health
Medications that have been suggested by doctors worldwide are available on the link below
https://mygenericpharmacy.com/category/products/blood-thinner