Warning Signs of Kidney Disease You Should Never Miss
Kidney Disease: Signs, Causes & Prevention Tips
Kidney disease often shows no early symptoms. Learn the warning signs, causes, and simple steps to protect your kidney health before it’s too late.
Kidney disease is often called a “silent killer“—and for good reason. The kidneys are remarkably resilient organs; they can function at a fraction of their capacity without causing noticeable pain or discomfort. By the time symptoms become obvious, significant and often irreversible damage has already occurred.
For a pharmacy audience, understanding these warning signs is critical. Pharmacists are often the most accessible healthcare professionals, seeing patients monthly for refills or weekly for minor ailments. Recognizing the subtle clues of kidney stress can lead to earlier intervention, potentially slowing disease progression and improving patient outcomes.
Why Early Detection Matters
Chronic Kidney Disease (CKD) affects an estimated 1 in 7 (15%) of U.S. adults, yet 9 in 10 adults with CKD do not know they have it. The danger lies in the progression. If caught early, lifestyle changes and medication can often halt or slow the damage. If missed, CKD can progress to kidney failure, requiring dialysis or a transplant.
Here are the warning signs you—and your patients—should never ignore.
1. Changes in Urination (The Earliest Clue)
Since the kidneys produce urine, changes in urinary habits are often the first red flag. These changes can be subtle and gradual, including:
- Frequency: Getting up more frequently at night to urinate (nocturia).
- Volume: Producing noticeably more or less urine than usual.
- Appearance: Urine that is foamy or bubbly (indicating protein, or albumin) or dark in color (indicating blood).
- Strain: Feeling pressure or having difficulty urinating.
Pharmacy Tip: When a patient mentions they are buying larger packs of incontinence pads, or cocomplainsbout interrupted sleep due to bathroom trips, it is worth asking about other risk factors like diabetes or hypertension.
2. Swelling (Edema) in Unexpected Places
When kidneys fail to filter properly, excess fluid and sodium build up in the body. This causes swelling, known as edema. It is most common in the:
- Hands and Feet: Patients may notice their shoes feel tight by the end of the day or that rings no longer fit.
- Ankles and Lower Legs: Pitting edema (pressure on the skin leaves an indent) is a classic sign.
- Face: Puffiness around the eyes, particularly in the morning, can indicate that the kidneys are leaking large amounts of protein.
Pharmacy Tip: Patients purchasing compression socks or diuretic herbal supplements (like dandelion or uva ursi) might be self-treating swelling. This is an opportunity to ask, “Have you mentioned this swelling to your doctor?” rather than just recommending a product.
3. Persistent Fatigue and “Brain Fog.”
Healthy kidneys produce a hormone called erythropoietin (EPO, which signals the body to make red blood cells. In kidney disease, EPO production drops, leading to anemia. This results in profound fatigue, weakness, and difficulty concentrating.
The Red Flag: Normal tiredness improves with rest. Kidney-related fatigue is a bone-deep weariness that sleep doesn’t fix. Patients may complain of feeling “foggy” or unable to focus, which is often mistaken for aging or stress.
4. Itchy Skin and Unexplained Rashes
When waste products build up in the blood (a condition called uremia), it can cause severe, persistent itching. This occurs because the kidneys are no longer able to balance minerals and nutrients, leading to high phosphorus levels, which trigger skin irritation.
The Red Flag: Patients may try over-the-counter antihistamines or hydrocortisone creams with little to no relief. If a patient presents with dry, itchy skin that doesn’t respond to standard treatments and they have risk factors (diabetes, high blood pressure), kidney function should be considered.
5. Metallic Taste and Ammonia Breath
The buildup of urea in the blood (uremia) can have a noticeable effect on the mouth. Patients may complain of:
- Ammonia Breath: Breath that smells like urine or ammonia.
- Metallic Taste: A persistent bad taste in the mouth or a strong aversion to eating meat (protein intolerance), as proteins break down and taste different to the affected patient.
- Weight Loss: This taste change can lead to a loss of appetite and unintended weight loss.
6. Muscle Cramps
Electrolyte imbalances—caused by failing kidneys—can lead to muscle cramps. Common culprits are low calcium levels and uncontrolled phosphorus. These cramps often occur in the legs or feet and can be disruptive to sleep.
Pharmacy Tip: While patients often reach for magnesium or potassium for cramps, those with kidney issues need to be very careful with electrolyte supplements, as their kidneys may not be able to filter the excess. Always ask about kidney health before recommending electrolyte replacements.
The Greatest Masquerader: No Symptoms at All
It is vital to remember that many people with moderate CKD (Stages 1-3) have zero symptoms. The only way to know for sure is through lab testing. The two key numbers are:
- eGFR (Estimated Glomerular Filtration Rate): A measure of kidney function. Below 60 for three months or more indicates CKD.
- Urine Albumin-to-Creatinine Ratio (UACR): Measures protein leaking into the urine.
High-Risk Populations (Who Needs to Be Extra Vigilant)
Certain populations are at higher risk and should be screened regularly, even without symptoms:
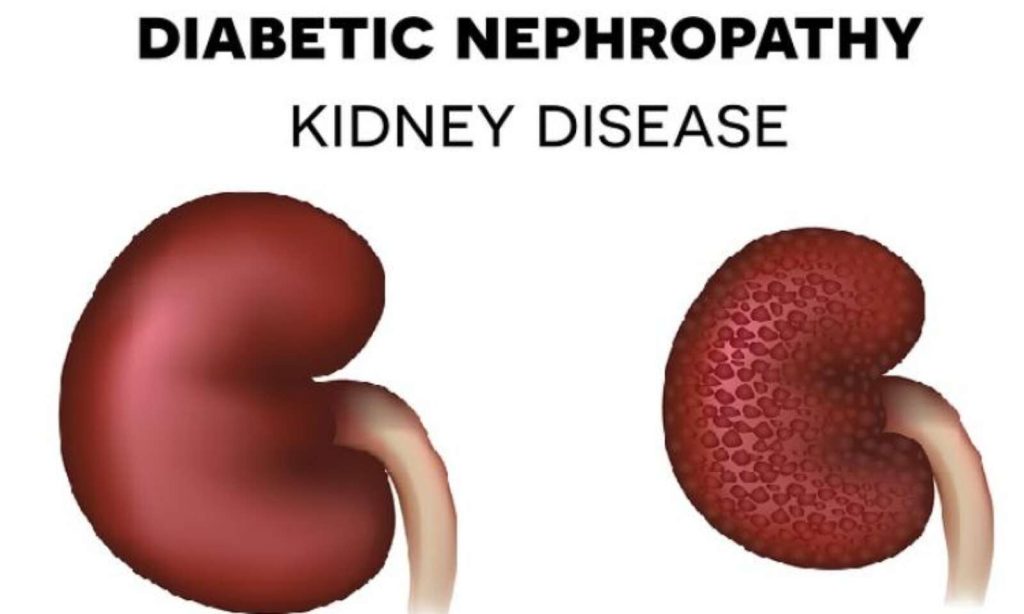
- Diabetics: Diabetes is the leading cause of kidney failure (responsible for nearly 40% of cases) .
- Hypertensives: High blood pressure is the second leading cause of kidney failure.
- Cardiovascular Disease: Heart disease and kidney disease often go hand-in-hand.
- Family History: Those with a family history of kidney failure or genetic conditions like Polycystic Kidney Disease (PKD).
- Long-term NSAID Use: Chronic use of ibuprofen or naproxen can damage the kidneys over time.
The Pharmacist’s Role: A Critical Safety Net
Pharmacists are uniquely positioned to spot kidney issues. Every time you process a prescription, you see the patient’s age and often their medication profile.
- Dose Adjustments: Are you verifying that medications (like Metformin, Gabapentin, or certain antibiotics) are dosed correctly for the patient’s kidney function? Many electronic systems now flag eGFR levels.
- OTC Counseling: Warn patients with hypertension or diabetes about the risks of NSAIDs. Steer them toward acetaminophen (Tylenol) for pain relief instead.
- Screening Opportunities: If a patient with diabetes picks up their meds, a simple question like, “When was the last time your doctor checked your kidney function?” can be a life-saving nudge.
Conclusion
Kidney disease is a progressive condition, but it doesn’t have to be a mystery. By recognizing the subtle signs—swollen ankles, foamy urine, persistent fatigue, and itchy skin—and understanding the critical lab values, pharmacists and patients alike can catch the disease early.
If you or a loved one is experiencing any of these warning signs, particularly if you have diabetes or high blood pressure, speak to a healthcare provider about getting a simple blood and urine test. Your kidneys are counting on you.
References for “Warning Signs of Kidney Disease”
https://www.cdc.gov/kidney-disease/php/data-research/index.html
https://www.kidney.org/kidney-topics/chronic-kidney-disease-ckd
https://diabetesjournals.org/care/article/47/Supplement_1/S219/153938/11-Chronic-Kidney-Disease-and-Risk-Management
https://kdigo.org/guidelines/ckd-evaluation-and-management/
https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/nonsteroidal-anti-inflammatory-drugs-nsaids
Medications that have been suggested by doctors worldwide are available on the link below
https://mygenericpharmacy.com/category/products/disease/chronic-kidney-disease
Disclaimer: This blog post is for informational purposes only and does not constitute medical advice. Always consult with a healthcare professional for medical advice, diagnosis, or treatment.