It’s a lifesaver when your immune system is functioning properly. That may be excellent, but it is not faultless. This unique collection of cells, tissues, and organs occasionally behaves improperly.
With autoimmune illnesses, your immune system attacks your body unintentionally. Rheumatoid arthritis, Crohn’s disease, and a few thyroid diseases are examples of these types.
What is an autoimmune disease?
Normally, the immune system protects against viruses and germs. It sends out an army of fighter cells to attack these foreign invaders as soon as it detects them.
The immune system can typically distinguish between your own cells and foreign cells. When you have an autoimmune disease, your immune system misinterprets your skin or joints as alien tissues. Autoantibodies, which are proteins released by the body, assault healthy cells.
Certain autoimmune disorders only affect a single organ. The pancreas is harmed by type 1 diabetes. Some illnesses, such as lupus or systemic lupus erythematosus (SLE), can have a total body impact.
A brief description of some of the most prevalent autoimmune illnesses is given below.
Why does the immune system attack the body?
Clinicians are unsure of the precise aetiology of immune system malfunction. Nonetheless, some people are more likely than others to develop an autoimmune disease.
According to a 2014 study, women are more likely than males to develop autoimmune illnesses (6.4% of women versus 2.7% of men). A lot of times, the illness strikes women who are fertile (ages 15 to 44).
Some ethnic groups are more likely to develop specific autoimmune illnesses. For instance, lupus affects white individuals less than it does African Americans and Hispanics.
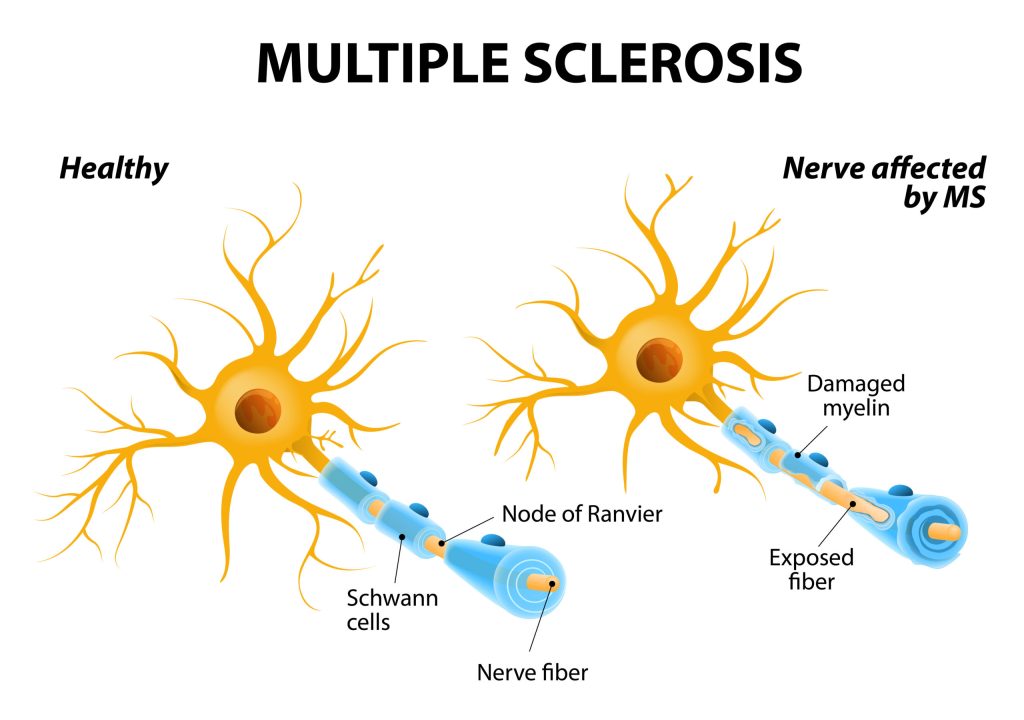
Many autoimmune conditions, including lupus and multiple sclerosis, run in families. Although not every family member will necessarily have the same illness, they all have a propensity for autoimmune diseases.
Researchers believe environmental factors like infections and exposure to chemicals or solvents may potentially play a role in the rise in the prevalence of autoimmune illnesses.
Another possible risk factor for developing an autoimmune illness is a “Western diet”. Consuming meals that are rich in fat, sugar, and processing is likely to contribute to inflammation, which may trigger an immunological response. But there is no proof of this.
SHORT VERDICT: The precise causation of autoimmune disorders is unknown. A number of factors may be at play, including genetics, nutrition, infections, and chemical exposure.
Your chance of contracting COVID-19 may increase if your immune system is compromised. If you have symptoms, make sure to get checked as soon as possible.
Common signs of a weak immune system
Remember that there are numerous additional reasons why these potential signals might appear. You should visit your doctor to find out what’s wrong with your health.
Cold hands
Your fingers, toes, ears, and nose may have a tougher time maintaining heat if your blood vessels are irritated. When exposed to cold, the skin in these places may turn white, then blue. The skin may turn red after the blood flow has resumed.
“Raynaud’s phenomenon,” as doctors refer to it. It can be brought on by immune system issues as well as by smoking, some prescription medications, and artery-related illnesses.
If you suffer from an autoimmune condition, your immune system is attacking your body rather than protecting it. Lupus and rheumatoid arthritis are two examples.
Dry eyes are a common symptom for those with autoimmune diseases. You can get a sand-like, grainy feeling in your eye. Astringent discharge, discomfort, redness, or blurred vision are some potential symptoms. Even when they are angry, some people find they are unable to cry.
Fatigue
Extreme fatigue similar to what you experience from the flu may indicate a problem with your body’s defences. Sleep probably won’t do any good. Your muscles or joints may also hurt. However, there may be a variety of different causes for your feelings.
Light Fever
Your immune system may be beginning to overwork itself if your body temperature is higher than usual. It may occur as a result of an impending infection or the beginning of an autoimmune disorder flare-up.
Headaches
Headaches can have an immune system component. Vasculitis, for instance, is the inflammation of a blood vessel brought on by an infection or an autoimmune disorder.
Rash
Your body’s first line of defence against pathogens is your skin. How it feels and looks may be an indication of how well your immune system is functioning.
Skin that is red, dry, and itchy is a typical sign of inflammation. The same goes for rashes that hurt or don’t go away. Lupus patients frequently develop a butterfly-shaped rash on their cheeks and nose.
Joint Pain
Your joints become sensitive to the touch when the lining inside them gets inflamed. It might affect more than one joint, and it may also be stiff or swollen. It can seem to be worse in the morning.
Patchy Hair Loss
The immune system can occasionally assault hair follicles. You can have a disorder called alopecia areata if you experience hair loss on your scalp, face, or other areas of your body. Hair breaking out in clumps or strands is another sign of lupus.
Continual Infections
Your body might not be able to effectively combat germs on its own if you need to take antibiotics more than twice per year (four times for kids).
Additional warning signs include persistent sinus infections, having more than four ear infections in a calendar year (for anybody older than 4), and recurring pneumonia.
Sun sensitivity
Photodermatitis, an allergic response to ultraviolet (UV) radiation, can occasionally occur in people with autoimmune diseases. After being in the sun, you can develop blisters, a rash, or scaly spots. Alternatively you can get nausea, a headache, or chills.
Numbness or Tingling in Your Feet and Hands
That might be entirely benign. Yet, in other circumstances it may indicate that your body is targeting the nerves that communicate with your muscles. For example, numbness that begins in the legs and spreads to the arms and chest may be a symptom of Guillain-Barre syndrome.
Acute inflammatory demyelinating polyneuropathy (AIDP), the demyelinating type of GBS, lasts for two to thirty days, whereas chronic inflammatory demyelinating polyneuropathy (CIDP) lasts longer. Longer-lasting is CIDP.
Difficulty swallowing
Your oesophagus, which transports food from your mouth to your stomach, may be enlarged or underdeveloped if you have trouble swallowing. Some individuals have a food-stuck sensation in their throat or chest. Those who swallow choke or gag. Your immune system could be one of the potential causes.
Unaccounted-for Weight Change
Even if your eating habits and exercise routine haven’t altered, you notice that you are putting on weight. Perhaps the number on your scale can fall without apparent cause. It’s possible that your thyroid gland has been harmed by an autoimmune disease as a result.
White Spots
Sometimes, melanocytes, the cells that produce colour in the skin, are targeted by the immune system. If so, your body will start to develop white patches of skin.
Your Skin or Eyes Are Turning Yellow
Jaundice, sometimes called biliary cirrhosis, is a condition where your immune system attacks and destroys healthy liver cells. This may result in autoimmune hepatitis, a disease.
Symptoms of autoimmune diseases
Several autoimmune illnesses have early signs and symptoms, including:
- fatigue
- stiff muscles
- swell and erythema
- minimal fever
- difficulty concentrating
- tingling and numbness in the hands and feet
- hair fall
- body rashes
Also, every disease may have a different set of symptoms. As an illustration, type 1 diabetes results in excessive thirst, weight loss, and exhaustion. IBD results in diarrhoea, bloating, and stomach pain.
Symptoms of autoimmune illnesses like psoriasis or RA can fluctuate. A flare-up is a time when symptoms are present. Remission refers to the time frame during which symptoms disappear.
VERDICT: Signs of an autoimmune disease may include weariness, muscle aches, swelling, and redness. With time, symptoms may appear and disappear.
REFERENCES:
For more details, kindly visit below.